Nomenclature (in this case, chemical nomenclature) is the term given to the naming of compounds. Since every chemical compound is different from one another, chemists use specific rules and conventions to name and identify them.
There are basically 3 types of compounds:
1. Ionic Compounds
2. Ionic Compounds With a Transition Metal
3. Covalent Compounds
Ionic compounds
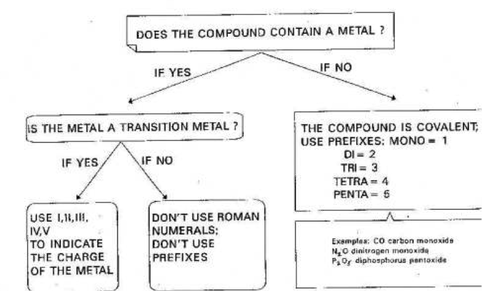
Ionic compounds are formed by a metal and a non-metal. Essentially, if the compound contains a metal, it is ionic. The following are two main rules that must be applied when naming ionic compounds.
· The metal ion's name does not change regardless of charge
· The non-metal's name ends in ide.
For example:
AlCl3 = aluminum chloride
Na2S = sodium sulfide
K2O = potassium oxide
MgH2 = magnesium hydride
*Notice how the little numbers (subscripts) do not affect the way we name the compounds.
Going backwards (name à formula): In such a case the total charge of the (+) and (-) ions in the compound must balance out.
Example: the formula of Calcium phosphide.
1. Consider the charges for Calcium and Phosphorus. In this case, Calcium has a charge of +2 and Phosphorus a charge of -3 (Ca+2 and P-3).
2. Balance out the charges. To do this, ask yourself, what number cam I use to give Ca and P the same number of charges (do not worry about positive and negative signs). In this case, 3 and 2 would suffice. (Ca(2)(3)=6 and P(3)(2)=6)
3. Put the numbers found in the previous step at the bottom of its respective element. These numbers become the subscripts.
Therefore the answer is: Ca3P2
*As you might have noticed, the numbers 3 and 2 crossed over (simply switched elements). In most cases, the “cross-over” method will work.
Other examples: Potassium oxide: K+1 and O-2 = K2O
Aluminum bromide: Al+3 and Br-1 = AlBr3
When metals are bonded to polyatomic ions the same rules apply, but you have to learn the names and charges of common polyatomic ions.
*Polyatomic ions = consist of two or more atoms with one overall charge.
Examples:
Aluminum sulfate: Al+3 and SO4-2 = Al2(SO4)3
*Notice that when there is more than one polyatomic group, we make use of brackets.
Here are a few examples of polyatomic ions:
Polyatomic Ion Name
OH-1 hydroxide
SO4-2 sulfate
PO4-3 phosphate
NO3-1 nitrate
CO3-2 carbonate
HCO3-1 hydrogen carbonate or bicarbonate
ClO3-1 chlorate
NH4+1 ammonium
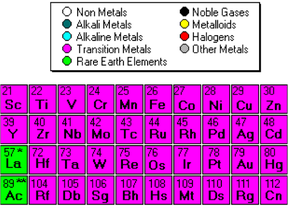
Ionic compounds with a transition Metal
These type of compounds are similar to ionic compounds; the only difference here is that there is a transition metal. In this case, we must specify the charge of the transition metal ion. To do this, we use a Roman numeral.
Roman numeral Charge
I +1
II +2
III +3
IV +4
V +5
VI +6
The image below shows which elements are considered to be transition metals.
There are basically 3 types of compounds:
1. Ionic Compounds
2. Ionic Compounds With a Transition Metal
3. Covalent Compounds
Ionic compounds
Ionic compounds are formed by a metal and a non-metal. Essentially, if the compound contains a metal, it is ionic. The following are two main rules that must be applied when naming ionic compounds.
· The metal ion's name does not change regardless of charge
· The non-metal's name ends in ide.
For example:
AlCl3 = aluminum chloride
Na2S = sodium sulfide
K2O = potassium oxide
MgH2 = magnesium hydride
*Notice how the little numbers (subscripts) do not affect the way we name the compounds.
Going backwards (name à formula): In such a case the total charge of the (+) and (-) ions in the compound must balance out.
Example: the formula of Calcium phosphide.
1. Consider the charges for Calcium and Phosphorus. In this case, Calcium has a charge of +2 and Phosphorus a charge of -3 (Ca+2 and P-3).
2. Balance out the charges. To do this, ask yourself, what number cam I use to give Ca and P the same number of charges (do not worry about positive and negative signs). In this case, 3 and 2 would suffice. (Ca(2)(3)=6 and P(3)(2)=6)
3. Put the numbers found in the previous step at the bottom of its respective element. These numbers become the subscripts.
Therefore the answer is: Ca3P2
*As you might have noticed, the numbers 3 and 2 crossed over (simply switched elements). In most cases, the “cross-over” method will work.
Other examples: Potassium oxide: K+1 and O-2 = K2O
Aluminum bromide: Al+3 and Br-1 = AlBr3
When metals are bonded to polyatomic ions the same rules apply, but you have to learn the names and charges of common polyatomic ions.
*Polyatomic ions = consist of two or more atoms with one overall charge.
Examples:
Aluminum sulfate: Al+3 and SO4-2 = Al2(SO4)3
*Notice that when there is more than one polyatomic group, we make use of brackets.
Here are a few examples of polyatomic ions:
Polyatomic Ion Name
OH-1 hydroxide
SO4-2 sulfate
PO4-3 phosphate
NO3-1 nitrate
CO3-2 carbonate
HCO3-1 hydrogen carbonate or bicarbonate
ClO3-1 chlorate
NH4+1 ammonium
Ionic compounds with a transition Metal
These type of compounds are similar to ionic compounds; the only difference here is that there is a transition metal. In this case, we must specify the charge of the transition metal ion. To do this, we use a Roman numeral.
Roman numeral Charge
I +1
II +2
III +3
IV +4
V +5
VI +6
The image below shows which elements are considered to be transition metals.
Examples:
· Manganese(II)oxide = Mn+2 and O-2. So we just need one of each and the formula becomes MnO.
· Copper(I) oxide à Cu+1 and O-2 which become Cu2O
Covalent compounds
These types of compounds are formed by a non-metal to a non-metal.
To name these compounds, we use a set of prefix to represent the subscript of an element.
Prefix Number of atoms
mono 1
di 2
tri 3
tetra 4
penta 5
hexa 6
hepta 7
octa 8
nona 9
deca 10
The only time we drop a prefix is if the mono is to appear at the beginning of the name.
Examples:
· CO = Carbon monoxide (note we don't say monocarbon monoxide)
· CO2 = Carbon dioxide
· Dinitrogen pentoxide = N2O5.
· Phosphorus trichloride PCl3
Summary